(Ch:37) Ultrasound Therapy
April 9, 2017How to Perform CEUS
May 4, 2017A case of prenatally diagnosed Binder syndrome
2nd Department of Obstetrics and Gynecology, Aristotle University of Thessaloniki, Thessaloniki, Greece
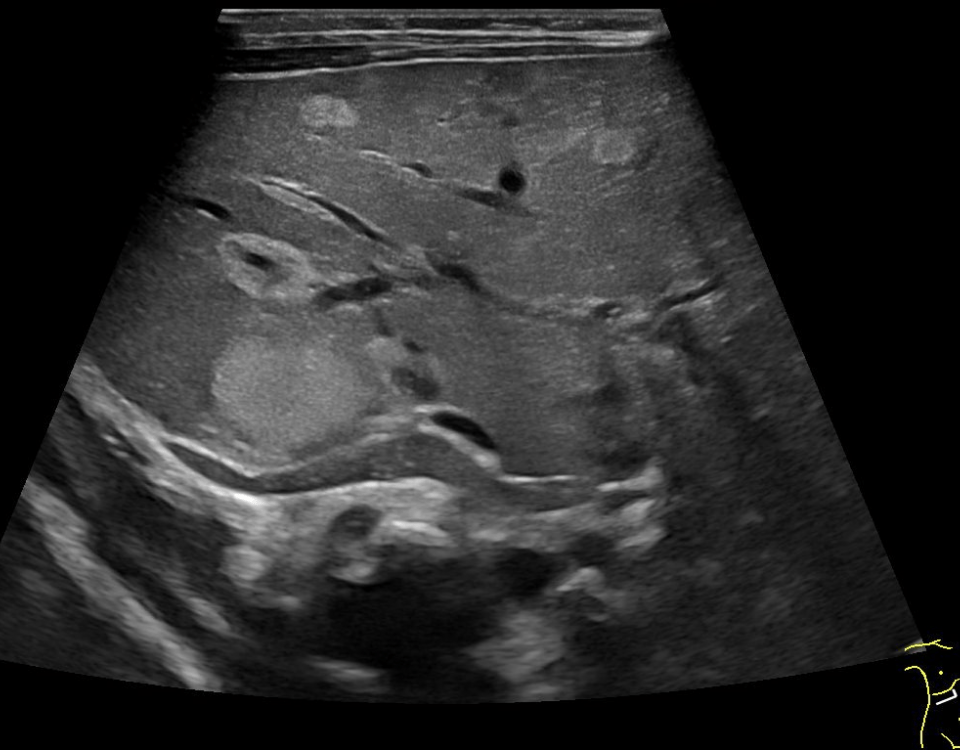
Binder syndrome (OMIM 155050, ORPHA 1248) is a rare condition characterized by midfacial hypoplasia, short nose and flat nasal bridge, underdeveloped upper jaw and a relatively protruding lower jaw (underbite) (http://www.orpha.net). Its inheritance pattern is unclear, but some recurrent cases may be explained by autosomal recessive inheritance with reduced penetrance or by multifactorial inheritance (1). Binder syndrome shares some common features with chondrodysplasia punctata (OMIM 215100) and it has been suggested that it may actually represent a mild form of the latter condition (2). Vitamin K deficiency has also been postulated as an environmental mechanism, resulting from maternal warfarin or phenytoin therapy, alcohol consumption, excessive vomiting, digestive bypass or an autoimmune disease (3-5). The exact prevalence of the condition is not known, but it may be in the realm of 1:10,000 (http://www.orpha.net). Prenatal detection of the Binder phenotype has been reported at the time of the second-trimester since 2000 (6), and it was based on the recognition of a characteristic facies with flattened nose in the midsagittal plane verticalized nasal bones and underbite (class III malocclusion).
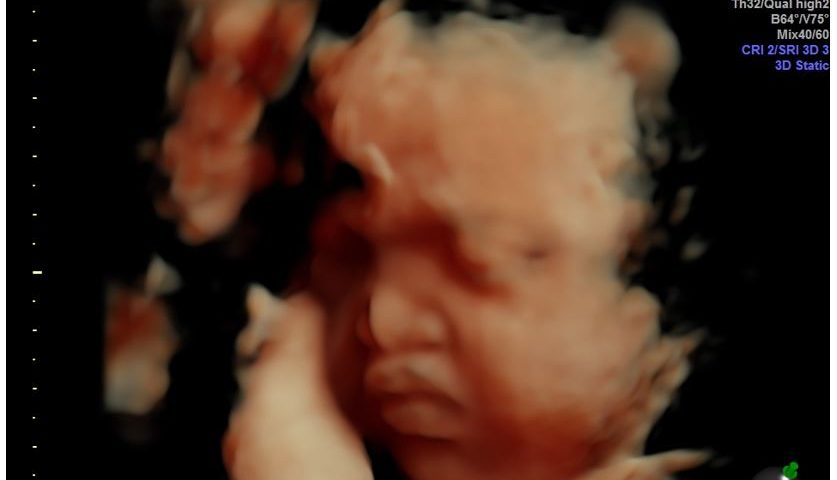
A 43-year old woman with 1 previous healthy child and a low-risk first-trimester screening test in the index pregnancy, presented at 21+4 weeks for routine second-trimester scan. At examination, a present (length 5.8 mm) but verticalized nasal bone was observed, resulting in a flat facial profile with underbite (Figures 1 and 2). No other major defects were detected and fetal biometry was observed. The couple were counselled about the tentative diagnosis of Binder syndrome, its potential implications and the limitations of prenatal diagnosis. Amniocentesis with array comparative hybridization (aCGH) was performed, indicating a normal female karyotype. At follow-up scans, no additional abnormalities were detected and the biometry of the long bones remained within normal limits. The course of pregnancy was uneventful, and an infant of 2750 g was born at 38+2 gestational weeks by elective caesarean section due to previous cesarean. At two months of life, the infant was doing well, and the only complaint at the time was tedious breathing and episodes of postprandial cough.
Ascertainment of diagnosis
Although the Binder profile is distinct, the lack of a recognized underlying chromosomal or molecular abnormality makes its prenatal ascertainment impossible. In a recent report of 8 cases with a prenatal Binder phenotype, one had fetal warfarin syndrome, one infantile sialic acid storage, one possible Keutel syndrome, and the other five had unclassifiable types of chondrodysplasia punctata (3). Although some of these conditions may have some additional sonographic features that can facilitate differential diagnosis (e.g. rhizomelic shortening of the long bones for autosomal recessive chondrodyspasia punctata), this is not always the case. It is therefore wise that detection of a Binder phenotype should trigger fetal karyotyping, fetal neurosonography or MRI (to exclude holoprosencephaly) and a helicoidal scan to detect stippling associated with chondrodysplasia punctata (3). Even so, the parents should be counseled that Binder syndrome is a tentative diagnosis prenatally, and that not all genetic syndromes can be prenatally excluded. Faced with this uncertainty, many parents would opt for pregnancy termination; among a total of 16 published cases with prenatal diagnosis, 6 (38%) chose for a termination (3, 6-10).
Prognosis
Hearing loss has been reported in 5% of Binder syndrome cases. The syndrome’s unique facial defects can be surgically corrected using a multi-stage multi-disciplinary approach which has a better prognosis if started early in life. Postponing surgical treatment results in a missed opportunity of normal-like facial growth and prolongation of the psychological burden, imposed by a negative self-image (11). Mental retardation, although reported in limited cases, it is not considered a significant feature of the syndrome (6).
It is important that antenatally diagnosed Binder Phenotype cannot definitely exclude more serious conditions, potentially involving neurodevelopmental delay. As noted before, out of eight cases of Binder phenotype antenatally diagnosed by Levaillant et al, two (25%) had an isolated Binder phenotype, while the remaining cases were related conditions sharing a similar phenotype.
Obstetric and perinatal care
No specific modification of the obstetric care is required. Recently, Blumenfeld et al reported three cases of antenatally diagnosed Binder phenotype associated to chondrodysplasia punctata, complicated by respiratory distress during the immediate post-natal period, attributed to obstruction of the narrowed upper airway (7). Therefore, care should be taken for preparation for neonatal resuscitation in such cases.
- Sheffield LJ, Halliday JL, Jensen F. Maxillonasal dysplasia (Binder's syndrome) and chondrodysplasia punctata. J Med Genet. 1991;28(7):503-4.
- Olow-Nordenram M, Valentin J. An etiologic study of maxillonasal dysplasia--Binder's syndrome. Scand J Dent Res. 1988;96(1):69-74.
- Levaillant JM, Moeglin D, Zouiten K, Bucourt M, Burglen L, Soupre V, et al. Binder phenotype: clinical and etiological heterogeneity of the so-called Binder maxillonasal dysplasia in prenatally diagnosed cases, and review of the literature. Prenat Diagn. 2009;29(2):140-50.
- Alessandri JL, Ramful D, Cuillier F. Binder phenotype and brachytelephalangic chondrodysplasia punctata secondary to maternal vitamin K deficiency. Clin Dysmorphol. 2010;19(2):85-7.
- Howe AM, Lipson AH, Sheffield LJ, Haan EA, Halliday JL, Jenson F, et al. Prenatal exposure to phenytoin, facial development, and a possible role for vitamin K. Am J Med Genet. 1995;58(3):238-44.
- Cook K, Prefumo F, Presti F, Homfray T, Campbell S. The prenatal diagnosis of Binder syndrome before 24 weeks of gestation: case report. Ultrasound Obstet Gynecol. 2000;16(6):578-81.
- Blumenfeld YJ, Davis AS, Hintz SR, Milan K, Messner AH, Barth RA, et al. Prenatally Diagnosed Cases of Binder Phenotype Complicated by Respiratory Distress in the Immediate Postnatal Period. J Ultrasound Med. 2016;35(6):1353-8.
- Boulet S, Dieterich K, Althuser M, Nugues F, Durand C, Charra C, et al. Brachytelephalangic chondrodysplasia punctata: prenatal diagnosis and postnatal outcome. Fetal Diagn Ther. 2010;28(3):186-90.
- Cantarell SM, Azuara LS, Perez SP, Juanos JL, Navarro FM, Martinez MC. Prenatal diagnosis of Binder's syndrome: report of two cases. Clin Exp Obstet Gynecol. 2016;43(2):279-83.
- Cuillier F, Cartault F, Lemaire P, Alessandri JL. Maxillo-nasal dysplasia (binder syndrome): antenatal discovery and implications. Fetal Diagn Ther. 2005;20(4):301-5.
- Monasterio FO, Molina F, McClintock JS. Nasal correction in Binder's syndrome: the evolution of a treatment plan. Aesthetic Plast Surg. 1997;21(5):299-308.
Figure 2: 3D reconstruction of the face in the same fetus at 32+4 gestational weeks


![A case of prenatally diagnosed Binder syndrome</br> [Apr 2017]](http://s834315022.websitehome.co.uk/wp-content/uploads/2020/11/cotm2017_apr-fig01a-b.jpg)
![A case of prenatally diagnosed Binder syndrome</br> [Apr 2017]](http://s834315022.websitehome.co.uk/wp-content/uploads/2020/11/cotm2017_april-fig02.jpg)