Student Image Challenge 98
October 11, 2023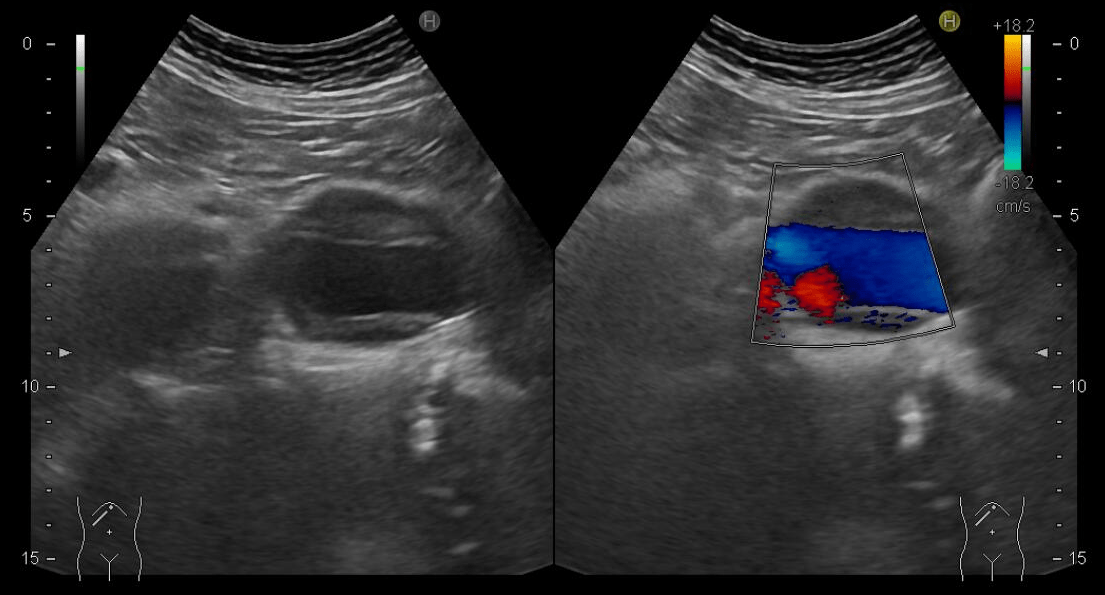
Student Image Challenge 99
November 16, 2023SUBMIT YOUR CASE
EFSUMB invites submission of interesting cases for the website section 'Case of the Month'. All CoM submissions are eligible for selection for free registration at the next Euroson congress. Two cases that receive the most 'likes' in a year will receive free registration for the next EUROSON congress and the third most liked liked case will receive a cash prize of 100 EUR.
2022 Winners were:
Top rated is Sonographic Diagnosis of a Pott’s Puffy Tumor in a 5-year-old-girl - [Jul 2022]
M Brandt Department of Otorhinolaryngology, Head and Neck Surgery, University Hospital of Regensburg, Germany
Second is Ischemic Colitis as Complication of SARS-CoV-2 Infection - [June 2022]
Federica Lepore Department of Internal Medicine, San Matteo Hospital Foundation, University of Pavia, Italy
Both winners received free registration at EUROSON2023.
View Submission Template
Giant hepatic cyst determining secondary Budd-Chiari syndrome: cruciality of ultrasound as a diagnostic and therapeutic tool
Authors: Simone Mammone[1], Antonio Cozzolino[2], Daniela Caterina Amoruso[2], Mario Romeo[1], Carmen D’Amore[3], Santolo Lo Sapio[4], Marina Cipullo[1], Marco Niosi[1], Marcello Dallio[1], Salvatore Scotto Di Santolo[2], Leonardo De Luca[2], and Alessandro Federico[1]
[1] Hepatogastroenterology Division, Department of Precision Medicine, University of Campania “Luigi Vanvitelli”, Naples, Italy[2] Gastroenterology Unit, Ospedale del Mare, Naples, Italy
[3] Cardiology Unit, Ospedale del Mare, Naples, Italy
[4] Intensive Care Unit, Ospedale del Mare, Naples, Italy
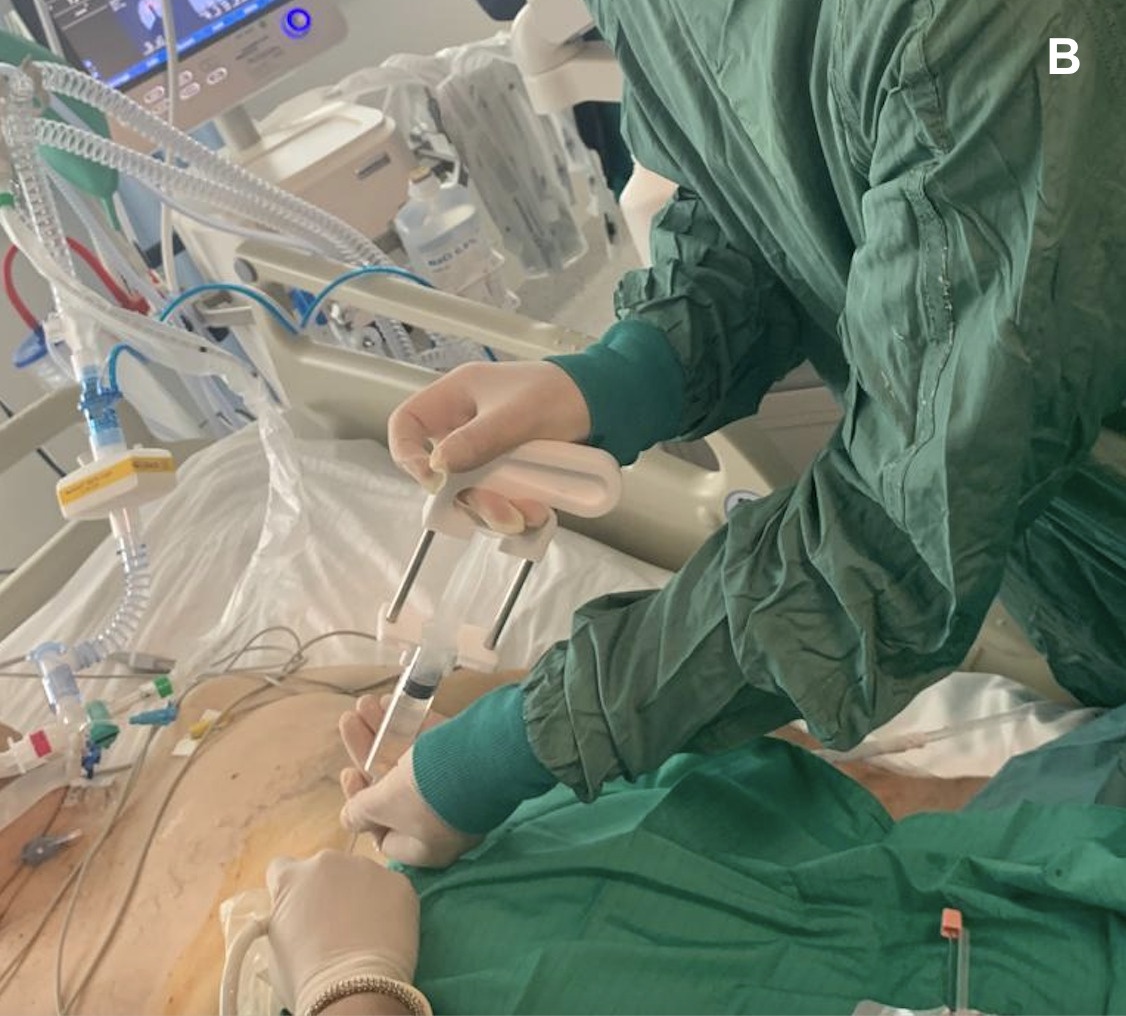
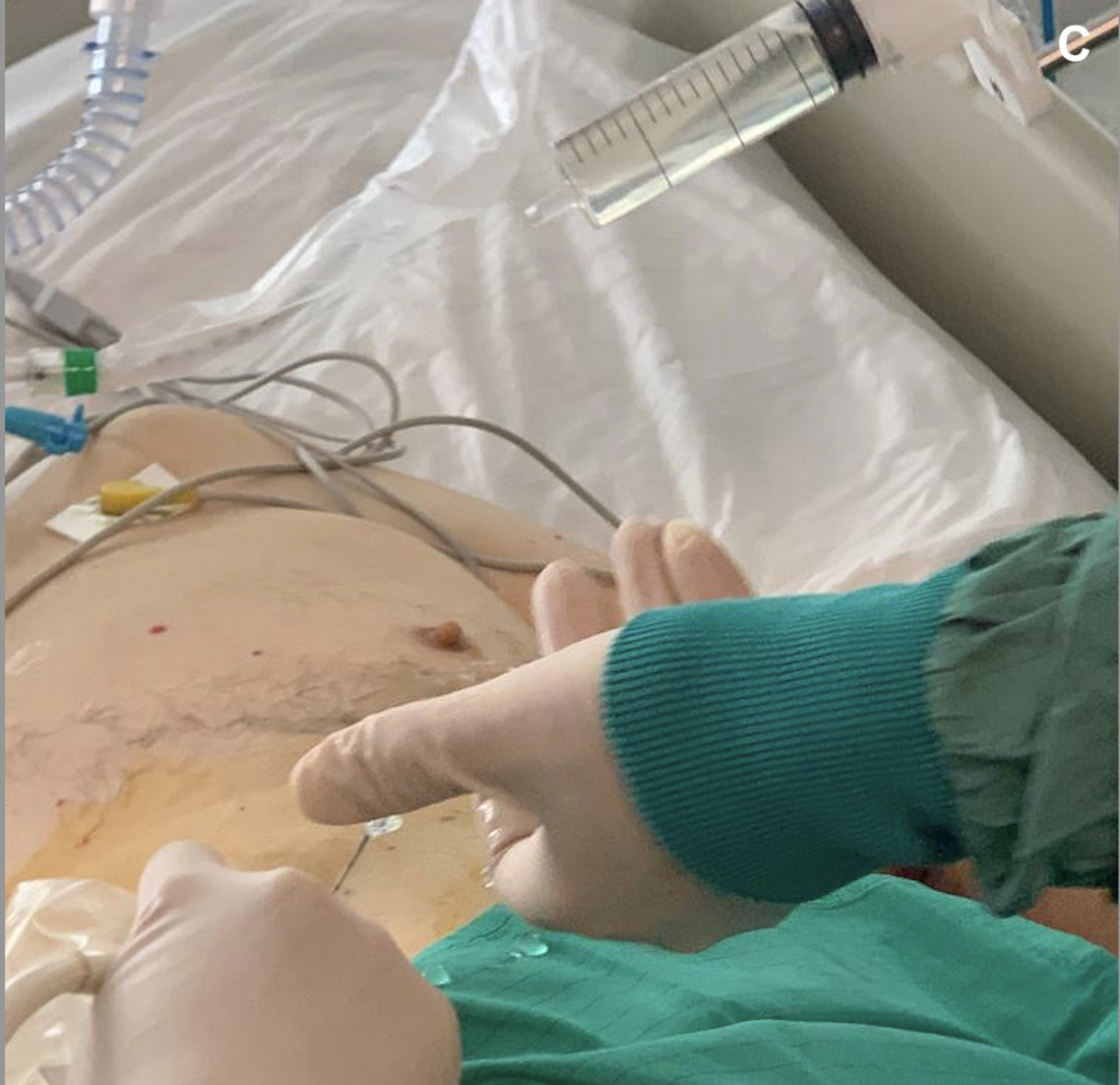
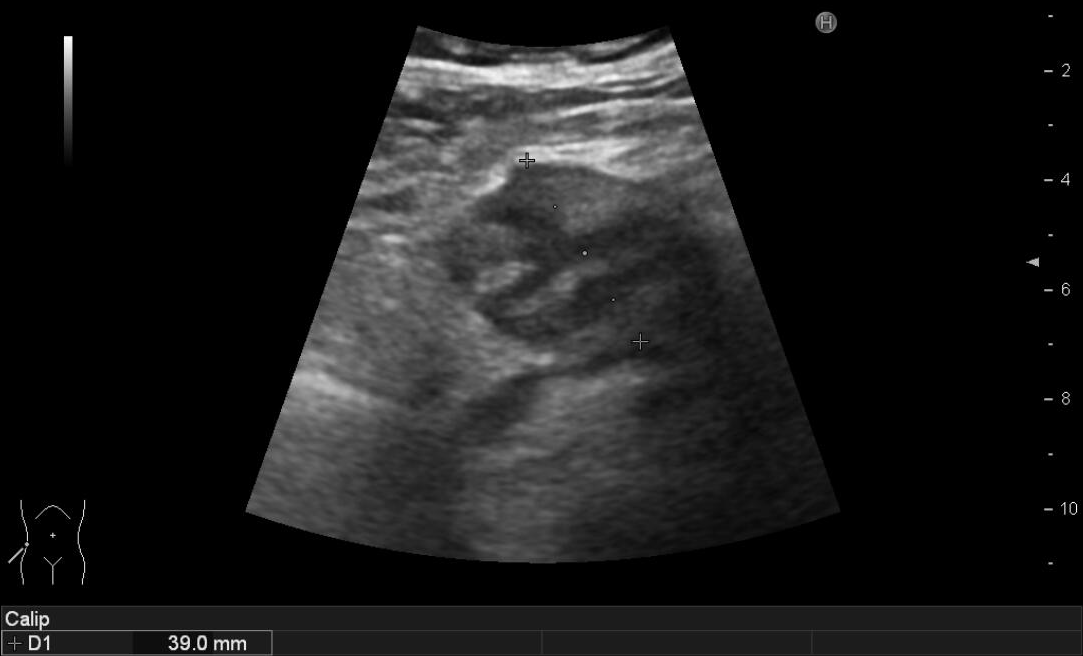
Figure 4A. Percutaneous aspiration of the hepatic cyst: after local anesthesia, we performed the US-guided cyst aspiration. In (A) introduction of an 18 Gauge x 20 cm Chiba needle connected to a syringe into the right hypochondrium (B-C), 150 mL of serum fluid was drained. The fluid tested negative on culture and cytological examination for tumor cells.
Figure 4B. Percutaneous aspiration of the hepatic cyst: after local anesthesia, we performed the US-guided cyst aspiration. In (A) introduction of an 18 Gauge x 20 cm Chiba needle connected to a syringe into the right hypochondrium (B-C), 150 mL of serum fluid was drained. The fluid tested negative on culture and cytological examination for tumor cells.
Figure 4C. Percutaneous aspiration of the hepatic cyst: after local anesthesia, we performed the US-guided cyst aspiration. In (A) introduction of an 18 Gauge x 20 cm Chiba needle connected to a syringe into the right hypochondrium (B-C), 150 mL of serum fluid was drained. The fluid tested negative on culture and cytological examination for tumor cells.
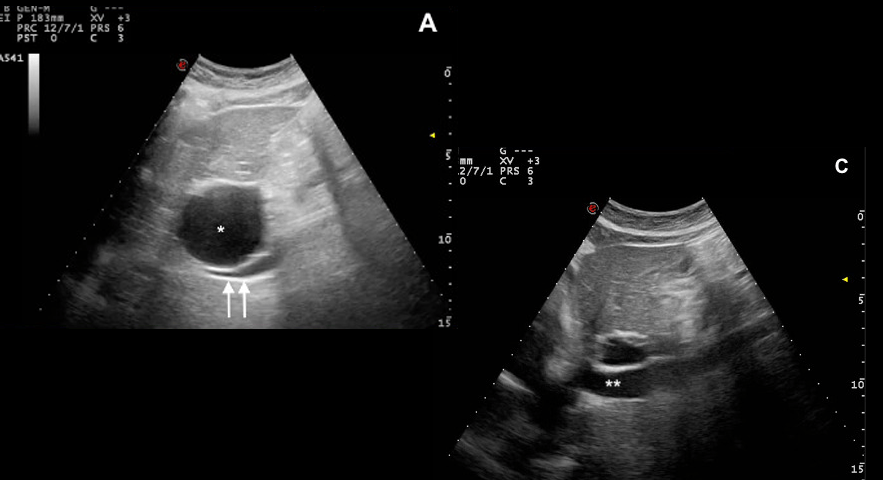
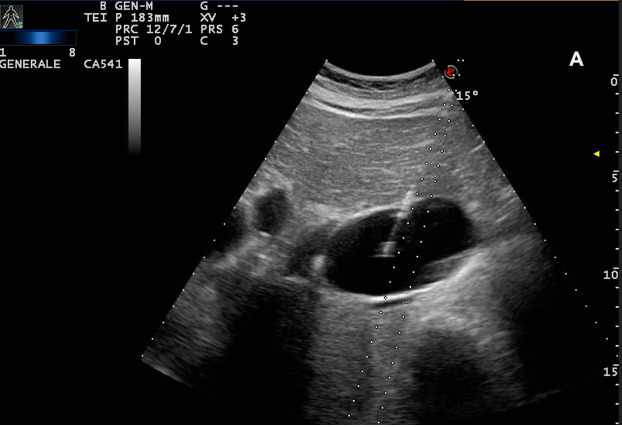
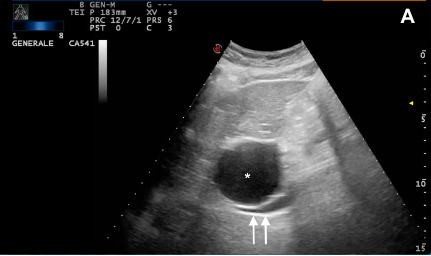
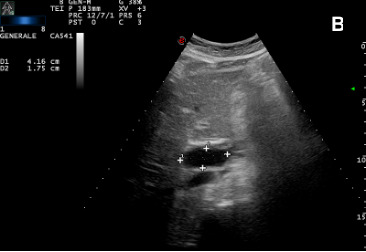
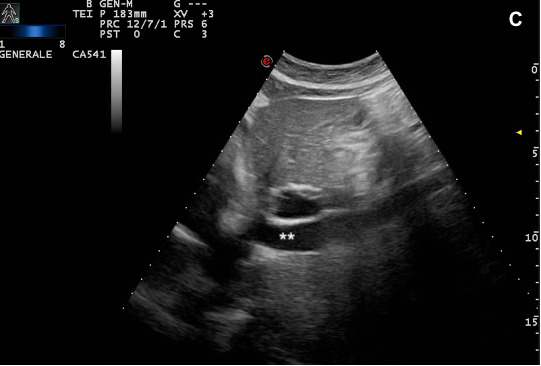
Figure 5A. Giant hepatic cyst (*) before (61x44 mm) (A) vs (14x17mm) (B) after the percutaneous aspiration. Restoration of adequate IVC caliber (**) (C). In our reported case, US proved to be an essential tool for both diagnostic and therapeutic purposes, providing a crucial management tool in an urgent scenario.
Figure 5B. Giant hepatic cyst (*) before (61x44 mm) (A) vs (14x17mm) (B) after the percutaneous aspiration. Restoration of adequate IVC caliber (**) (C). In our reported case, US proved to be an essential tool for both diagnostic and therapeutic purposes, providing a crucial management tool in an urgent scenario.
Figure 5C. Giant hepatic cyst (*) before (61x44 mm) (A) vs (14x17mm) (B) after the percutaneous aspiration. Restoration of adequate IVC caliber (**) (C). In our reported case, US proved to be an essential tool for both diagnostic and therapeutic purposes, providing a crucial management tool in an urgent scenario.
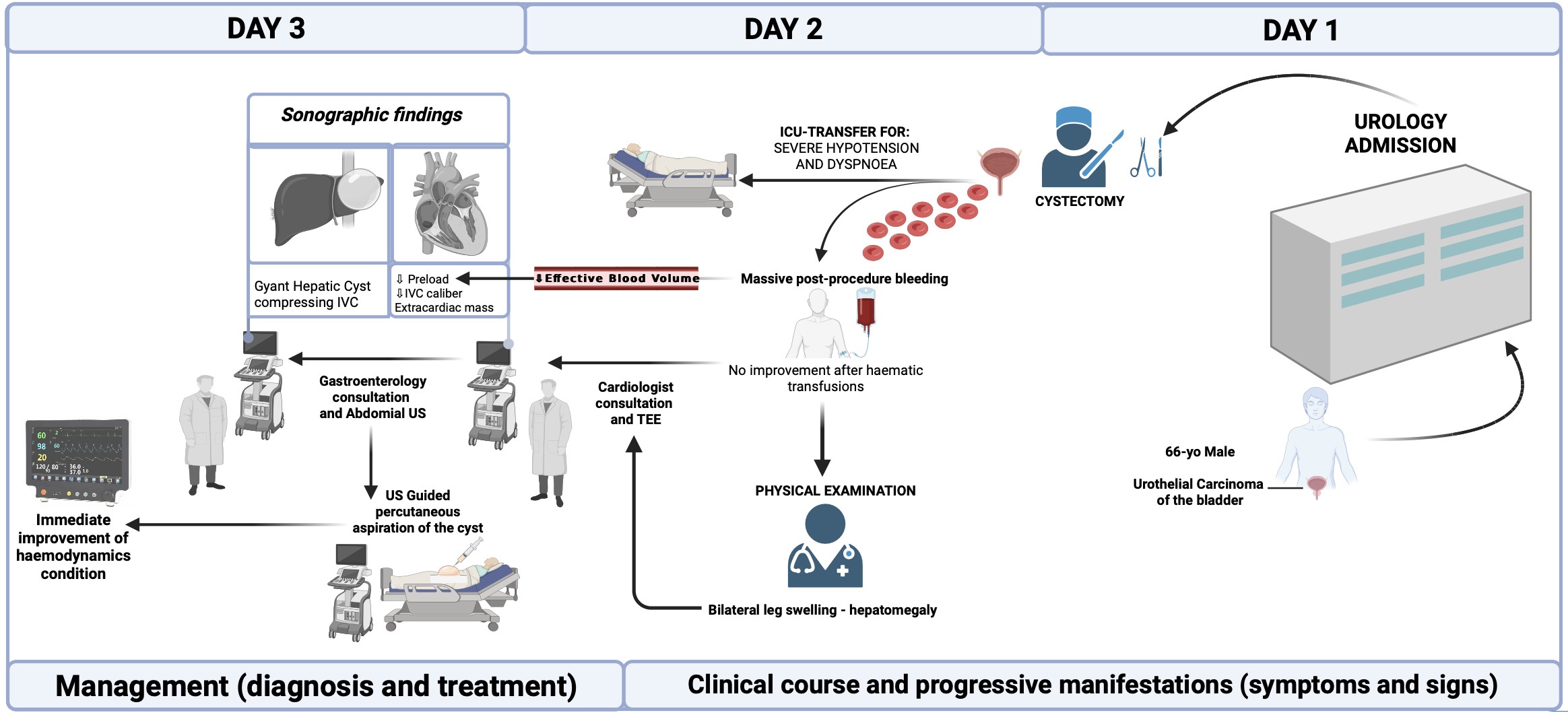
The timeline of clinical events is reported in Figure 1.
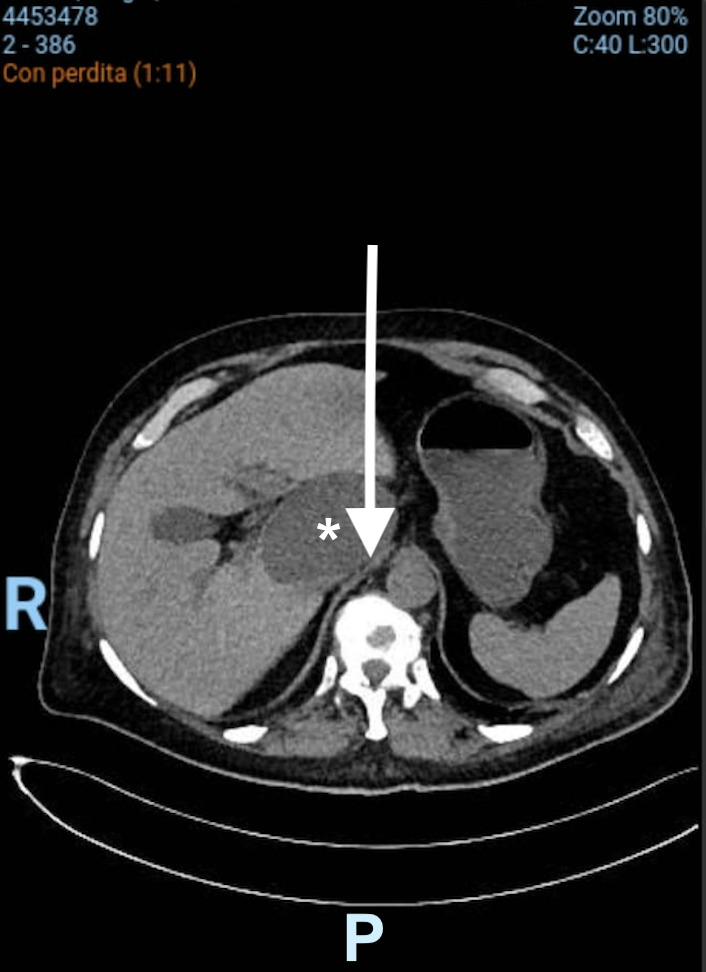
Computed Tomography (CT) subsequently confirmed the US diagnosis, revealing a hypodense nodular hepatic lesion compatible with a giant (62 x 55 mm) cyst compressing the IVC and the absence of active bleeding (Figure 3).
Altogether, these findings allowed the diagnosis of a giant hepatic simple cyst compressing the IVC, thus configuring the clinical scenario of “secondary Budd-Chiari syndrome” (sBCS) or “inferior vena cava syndrome” (IVCs). In particular, it appeared highly likely that the compressive effect of this lesion, which probably developed in the span of several years and had a gradual increase in size, has led to a hemodynamically compensated state. However, post-procedural bleeding represented the trigger that led to hemodynamic decompensation by determining a reduction in effective blood volume.
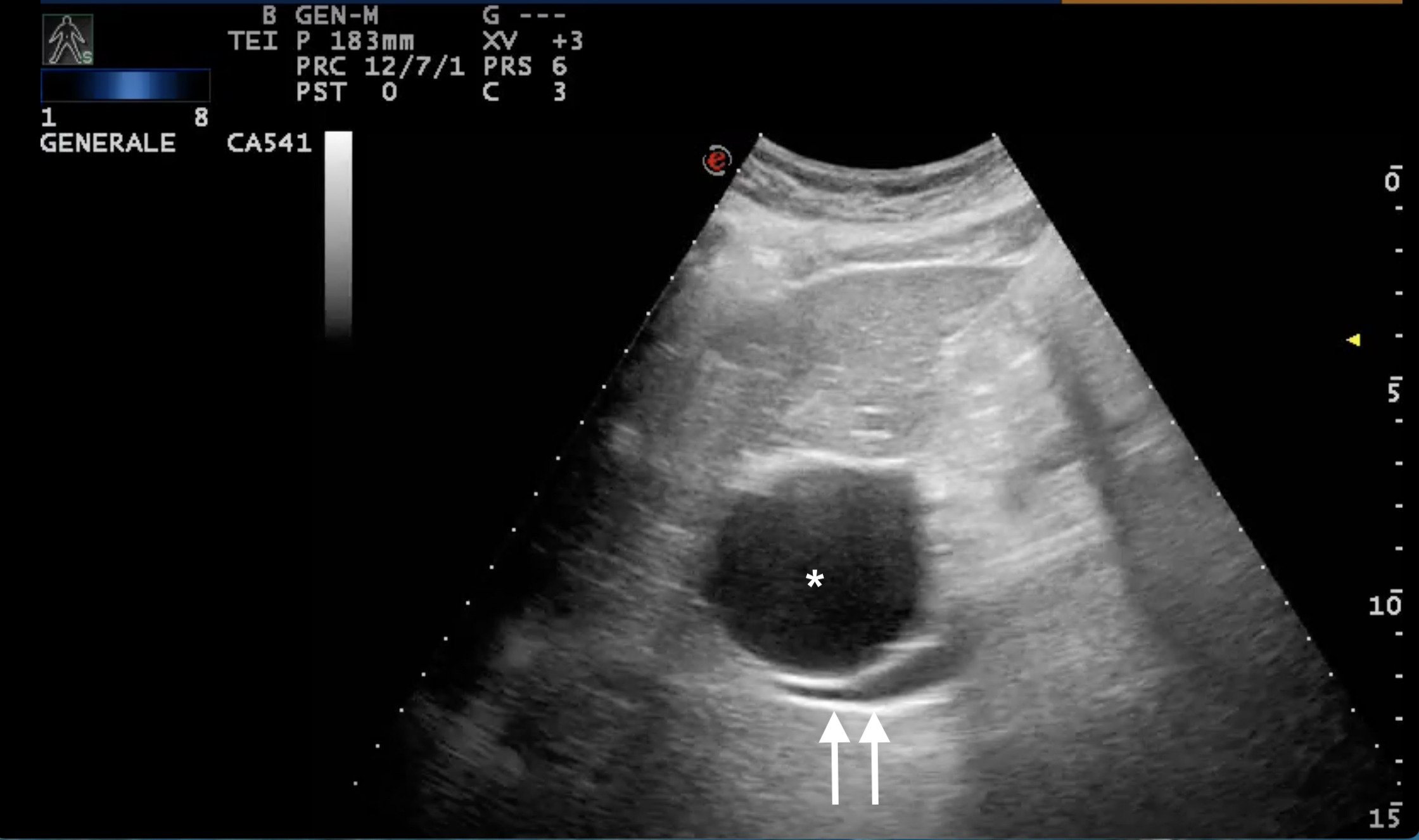
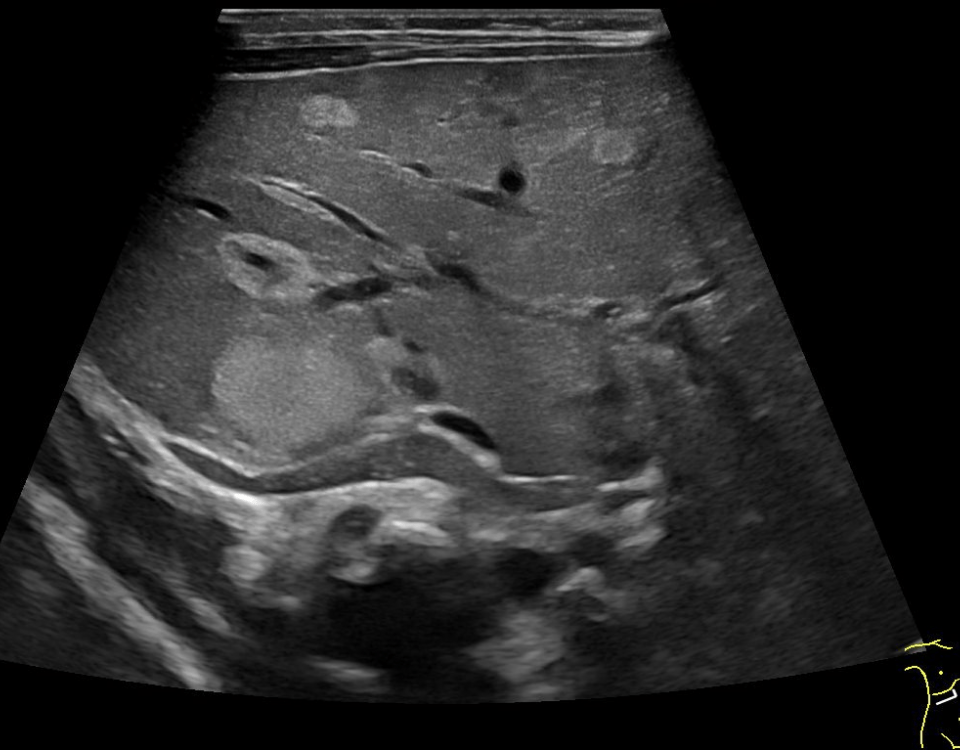
Figure 2. B-scan ultrasound: giant (61x44 mm) hepatic cyst (*) localized at I-IIs significantly compressing the IVC (double arrow)
Figure 3. CT-scan: giant (62x55 mm) hepatic cyst (*) localized at I-IIs significantly compressing (arrow) the IVC.
In light of this, considering the rapid deterioration of the patient’s clinical conditions and given that the patient was unfit for surgery, we decided to perform an urgent ultrasound-guided percutaneous transhepatic aspiration of the cyst (Figure 4 A, B, C)
Figure 4. Percutaneous aspiration of the hepatic cyst: after local anesthesia, we performed the US-guided cyst aspiration. In (A) introduction of an 18 Gauge x 20 cm Chiba needle connected to a syringe into the right hypochondrium (B-C), 150 mL of serum fluid was drained. The fluid tested negative on culture and cytological examination for tumor cells.
Contextually, a significant decrease in cyst dimension (14x17mm) (Figures 5A and B) and, more relevant, a reduction of the IVC compression (Figure 5C) led to the rapid restoration of the hemodynamic condition, stabilizing the patient at a final blood pressure of 122/60 mmHg Consequently, the clinical success has shown that this procedure represented a life-saving approach for the patient in this hyperacute scenario. We reserved the window of opportunity that, depending on the rapidity of recurrence of the lesion, to perform alternative approaches, which might entail additional procedural-related risks, including ethanol cyst sclerotherapy or IVC stenting.
Figure 5. Giant hepatic cyst (*) before (61x44 mm) (A) vs (14x17mm) (B) after the percutaneous aspiration. Restoration of adequate IVC caliber (**) (C). In our reported case, US proved to be an essential tool for both diagnostic and therapeutic purposes, providing a crucial management tool in an urgent scenario.
Budd-Chiari syndrome is an uncommon vascular disorder caused by hepatic venous flow obstruction determining an impaired venous outflow from the liver, consequently leading to an increase in sinusoidal and portal pressures and a decrease in portal flow. The obstruction may be localized along any point of the venous course, from the hepatic venules to the right atrium. Pathologically, BCS can be subdivided into primary (pBCS), when intrinsic intraluminal thrombosis and stenosis determine the flow obstruction, or secondary (sBCS) when a malignant (tumor) or benign (abscesses, cysts) mass determine an ab extrinsic compression of the hepatic veins or inferior vena cava (“IVC syndrome”). According to the clinical course, BSC can be classified into fulminant, acute, subacute, and chronic (1). Worldwide, pBCS frequency is more frequent than sBCS and is estimated at 1/1000000 inhabitants/year. Etiological factors include congenital abnormalities, hypercoagulable conditions, inferior vena cava anatomical variability, myeloproliferative diseases, and environmental conditions.
CLINICAL PERSPECTIVE:
BCS may vary from a complete asymptomatic condition to fulminant liver failure. Symptom severity varies depending on the underlying mechanism of the impaired venous outflow. The most common symptoms are abdominal pain, ascites, hepatomegaly, lower limb swelling, and jaundice. BCS can often be underdiagnosed in clinical practice since up to 20% of cases are asymptomatic, indicating a chronic onset of hepatic venous obstruction (2). In the case of IVC syndrome, the systemic hypotension with generalized manifestations of hypoperfusion (e.g., dyspnea, anuria) may represent a common epiphenomenon when the reduction of cardiac output, consequent to the decreased preload, becomes hemodynamically significant (2). The diagnosis is established by demonstrating the hepatic venous outflow tract obstruction, usually by non-invasive imaging. Indeed, irrespective of the cause, Doppler abdominal ultrasonography is the key diagnostic tool of BCS (3). CT and magnetic resonance imaging (MRI) represent second-line tests and are limited due to their uncertain results in approximately 50% of cases (4).
THERAPY PLANNING:
The therapeutic approach to BCS depends on the cause and should be determined on an individual basis. Concerning pBCS, the first step is represented by medical treatment (anticoagulation therapy or thrombolysis), but it may not be sufficient in several patients. The second-line therapy for patients with short-length stenosis is angioplasty or stenting. The next step is the transjugular intrahepatic portosystemic shunt (TIPS), which decompresses the splanchnic circulation. The last step is represented by liver transplantation (LT). About the secondary form, surgery is usually the best option. This treatment aims to restore hepatic venous drainage and thereby decompress the liver and, possibly, the IVC. This is usually done through the removal of extrinsic compression or by positioning a stent in the IVC lumen. However, in the case of liver cysts, aspiration (eventually followed by sclerotherapy) is the preferred treatment, in particular in the cases with acute onset, which lead to a rapid deterioration of the patient's status. More definitive therapeutic options include laparoscopic or open cyst fenestration, which mandates resection of the cyst roof or, rarely, hepatic resection (6).
OUTCOME & PROGNOSIS:
In the absence of adequate treatment, the natural course of BCS leads to liver failure. Usually, medium-term prognosis depends on the severity of the liver disease, according to the model for end-stage liver disease (MELD) score or Child-Pugh. Early TIPS proved to improve BCS outcomes. In patients treated for BCS, the most common causes of death are liver failure, postoperative multiorgan failure, and sepsis (5). When sBCS is configured by IVC syndrome, the hepatic involvement can occur at a later stage in the disease course and, in most cases, remains unobserved given the predominance of systemic hemodynamic dysfunction which directy impacts short- and long-term outcomes if not promptly addressed.
2. Tomáš G, Lukáš L, Gabriela G, et al.. Budd-Chiari Syndrome. Prague Medical Report 2017; 118: 69–80
3. Aydinli M, Bayraktar Y. Budd-Chiari syndrome: etiology, pathogenesis and diagnosis. World Gastroenterol 2007; 13:2693–2696
4. Iliescu L, Toma L, Mercan-Stanciu A, et al.. S. Budd-Chiari syndrome - various etiologies and imagistic findings. A pictorial review. Med Ultrason. 2019; 21: 344-348.
5. Murad, S. D., Valla, D.-C., de Groen, P. C., et al.. Determinants of survival and the effect of portosystemic shunting in patients with Budd-Chiari syndrome. Hepatology 2004; 39: 500-8.
6. Reid-Lombardo KM, Khan S, Sclabas G. Hepatic cysts and liver abscess. Surg Clin North Am. 2010; 90: 679-97