Sonographic Diagnosis of a Pott’s Puffy Tumor in a 5-year-old-girl [Jul 2022]
August 4, 2022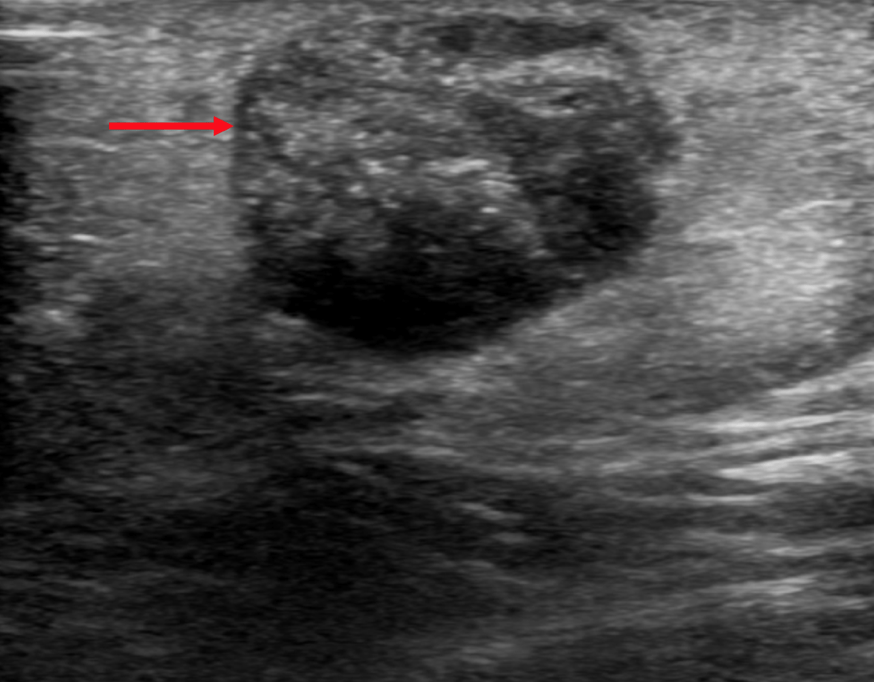
Pilomatricoma: A Case Report with Pathological Confirmation [Sept 2022]
November 1, 2022MALT Lymphoma developed on a Sjogren Syndrome
AUTHORS
Manuela Lenghel¹,
Raluca Petea-Balea¹,
Laura Damian²,
Cristian Dinu³,
Georgeta Rusu¹,
Csaba Csutak¹,
Maria Badaranza⁴
[1] Department of Radiology, “Iuliu Hațieganu” University of Medicine and Pharmacy, 400012 Cluj-Napoca, Romania
[2] Department of Rheumatology, Center for Rare Autoimmune and Autoinflammatory Diseases, Emergency Clinical County Hospital Cluj, 2-4 Clinicilor St, 400006 Cluj-Napoca, Romania
[3] Department of Maxillofacial Surgery and Radiology, Maxillofacial Surgery and Implantology, “Iuliu Hațieganu” University of Medicine and Pharmacy, 37 Iuliu Hossu Street, 400429 Cluj-Napoca, Romania
[4] 2(nd) Department of Internal Medicine, Iuliu Hatieganu University of Medicine and Pharmacy, Cluj-Napoca, Romania.
Manuela Lenghel¹,
Raluca Petea-Balea¹,
Laura Damian²,
Cristian Dinu³,
Georgeta Rusu¹,
Csaba Csutak¹,
Maria Badaranza⁴
[1] Department of Radiology, “Iuliu Hațieganu” University of Medicine and Pharmacy, 400012 Cluj-Napoca, Romania
[2] Department of Rheumatology, Center for Rare Autoimmune and Autoinflammatory Diseases, Emergency Clinical County Hospital Cluj, 2-4 Clinicilor St, 400006 Cluj-Napoca, Romania
[3] Department of Maxillofacial Surgery and Radiology, Maxillofacial Surgery and Implantology, “Iuliu Hațieganu” University of Medicine and Pharmacy, 37 Iuliu Hossu Street, 400429 Cluj-Napoca, Romania
[4] 2(nd) Department of Internal Medicine, Iuliu Hatieganu University of Medicine and Pharmacy, Cluj-Napoca, Romania.
1Clinical History
We present the case of a 42-year-old female patient, diagnosed with primary Sjogren syndrome, with bilateral involvement of the major salivary glands (parotid and submandibular). She was treated with symptomatic agents and Hydroxychloroquine. Ten years after diagnosis, she developed a painless nodular swelling in the left parotid gland. Laboratory showed lymphopenia and inconstantly urinary monoclonal free lambda chains.
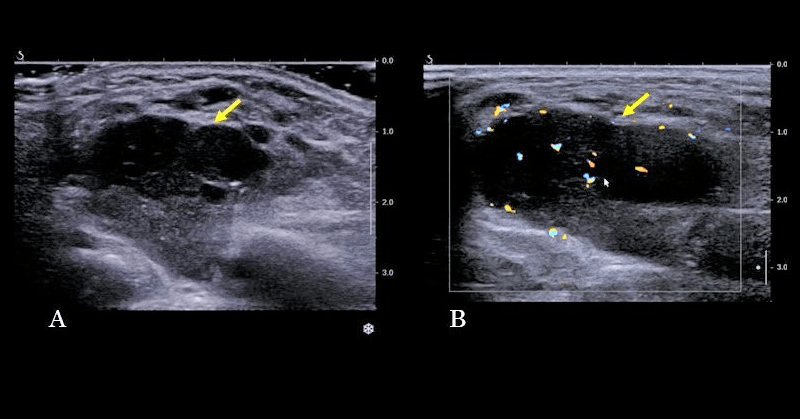
Ultrasound revealed a nodular, inhomogeneous hypoechoic area with echogenic stripes suggesting parotid lymphoma complication ( Figure 1). Although the parotid biopsy did not show malignant transformation, the nodular area was surgically removed two months later. The histopathological exam revealed a benign lymphoepithelial lesion. Azathioprine immunosuppression was added to her treatment. Five years after the initial surgical intervention, she presented again with a swelling of the left parotid gland. Laboratory showed positive antinuclear antibodies with negative anti-dsDNA, positive anti-Ro and anti-LA antibodies, and positive rheumatoid factor. She had negative cryoglobulins, normal C3, C4, and immunoglobulins, but with the presence of a monoclonal gammopathy.
Ultrasound revealed a nodular, inhomogeneous hypoechoic area with echogenic stripes suggesting parotid lymphoma complication ( Figure 1). Although the parotid biopsy did not show malignant transformation, the nodular area was surgically removed two months later. The histopathological exam revealed a benign lymphoepithelial lesion. Azathioprine immunosuppression was added to her treatment. Five years after the initial surgical intervention, she presented again with a swelling of the left parotid gland. Laboratory showed positive antinuclear antibodies with negative anti-dsDNA, positive anti-Ro and anti-LA antibodies, and positive rheumatoid factor. She had negative cryoglobulins, normal C3, C4, and immunoglobulins, but with the presence of a monoclonal gammopathy.
2Image findings
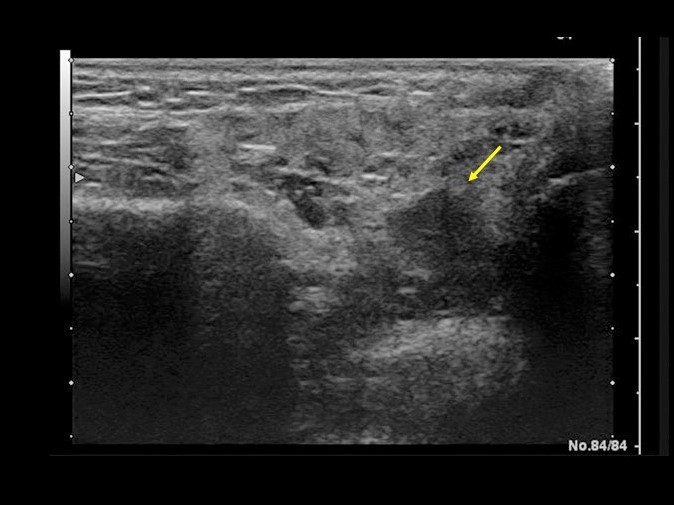
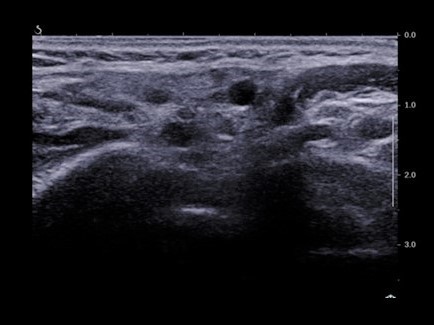
An ultrasound examination was performed, and, besides the architectural changes in the salivary glands parenchyma suggestive of Sjogren Syndrome, in the left parotid gland a nodular lesion was detected. The lesion was inhomogeneous, hypoechoic, with Doppler signal, but with irregular distribution of the vessels (Figure 2 and Figure 3).
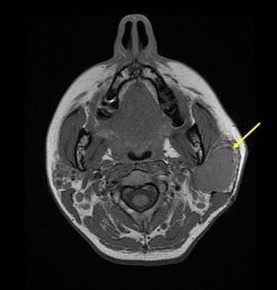
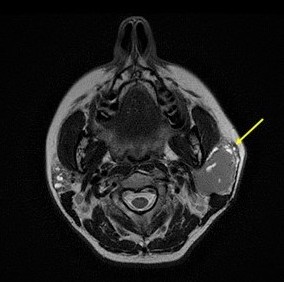
An MRI examination completed the ultrasound evaluation, revealing a decreased signal intensity on T1 and T2 sequences, with an area of high signal on T2 images (Figure 5, Figure 6).
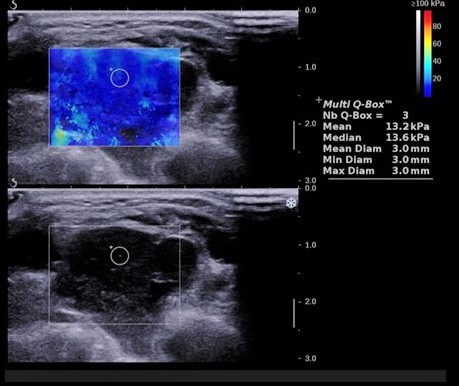
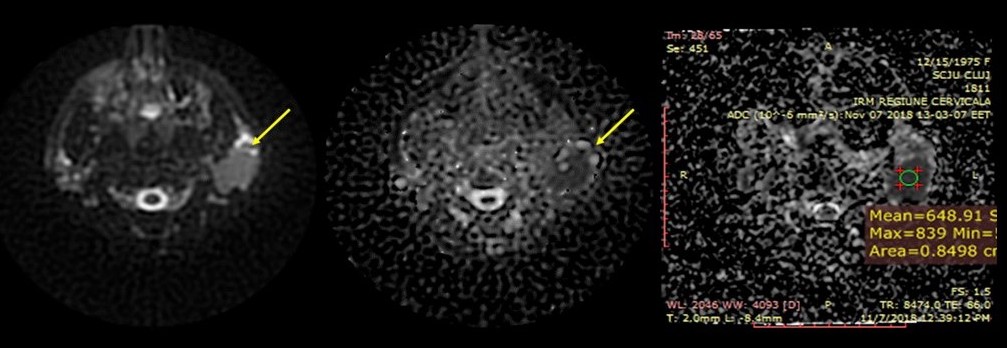
On diffusion weight imaging (DWI) sequences an important diffusion restriction was shown, with an apparent diffusion coefficient (ADC) value of 0.64 x 10-3 mm2/sec (Figure 7).
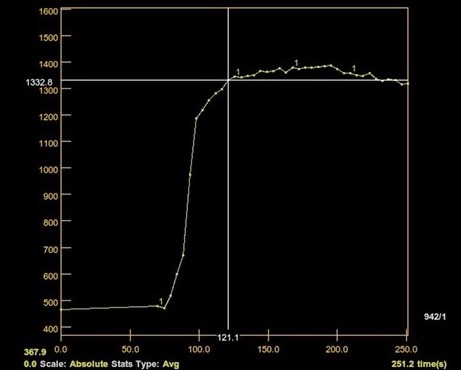
Dynamic Contrast-Enhanced (DCE) MRI showed a type B curve, with progressive enhancement, followed by a plateau phase and a wash-out (Figure 8).
Based on the DCE-MRI findings was raised the suspicion of lymphomatous degeneration and the patient was referred to the surgical department for excisional biopsy. The histopathological exam confirmed the presence of extranodal marginal zone B-cell lymphoma left parotid gland.
An MRI examination completed the ultrasound evaluation, revealing a decreased signal intensity on T1 and T2 sequences, with an area of high signal on T2 images (Figure 5, Figure 6).
On diffusion weight imaging (DWI) sequences an important diffusion restriction was shown, with an apparent diffusion coefficient (ADC) value of 0.64 x 10-3 mm2/sec (Figure 7).
Dynamic Contrast-Enhanced (DCE) MRI showed a type B curve, with progressive enhancement, followed by a plateau phase and a wash-out (Figure 8).
Based on the DCE-MRI findings was raised the suspicion of lymphomatous degeneration and the patient was referred to the surgical department for excisional biopsy. The histopathological exam confirmed the presence of extranodal marginal zone B-cell lymphoma left parotid gland.
3Diagnosis
Final diagnosis: MALT lymphoma (Extranodal marginal zone B-cell lymphoma) of the left parotid gland in a patient with overlap syndrome (Sjogren Syndrome and subacute cutaneous lupus).
4Discussion
BACKGROUND
Patients with Sjogren Syndrome have an increased risk of developing non-Hodgkin lymphoma (especially MALT lymphoma). Based on the long history of Sicca symptoms (keratoconjunctivitis sicca and xerostomia) associated with the appearance of a nodular lesion in the parotid gland, a suspicion of MALT lymphoma should be raised [1].
CLINICAL PERSPECTIVE
The challenge was represented by the presence of a unique, unilateral lesion of the left parotid gland, which proved to be a benign lymphoepithelial lesion (BLEL) associated with Sjogren syndrome preceding the appearance of the MALT lymphoma [1]. Differential diagnosis: Tumor-like benign lymphoepithelial lesion (BLEL), also known as lymphoepithelial sialadenitis associated with Sjogren syndrome; HIV-related benign lymphoepithelial lesion [1].
THERAPY PLANNING:
Differentiation between BLEL and MALT lymphoma is often difficult, but nowadays can be made using Dynamic Contrast-Enhanced MRI (for BLEL, time to peak is more than twice compared with MALT lymphoma) and Diffusion-weighted MRI (ADC values are lower in case of BLEL, compared to MALT lymphoma). Differentiation from HIV-related benign lymphoepithelial lesions is based on the fact that solid masses of the parotid gland are always associated with cystic masses, tonsillar hyperplasia, and cervical reactive adenopathy [2,3].
OUTCOME:
Lymphomas associated with Sjogren Syndrome often present as an indolent lymphoma with rare involvement of the bone marrow and absence of B symptoms. Only in 10% of the cases, low-grade lymphomas can transform into high-grade lymphoma, most frequently Diffuse Large B-Cell Lymphoma [4].
PROGNOSIS:
MALT lymphoma associated with Sjogren Syndrome tends to have a good prognosis and can be cured with surgical excision. When surgical treatment is not an option, chemotherapy and radiotherapy can be considered [5].
Patients with Sjogren Syndrome have an increased risk of developing non-Hodgkin lymphoma (especially MALT lymphoma). Based on the long history of Sicca symptoms (keratoconjunctivitis sicca and xerostomia) associated with the appearance of a nodular lesion in the parotid gland, a suspicion of MALT lymphoma should be raised [1].
CLINICAL PERSPECTIVE
The challenge was represented by the presence of a unique, unilateral lesion of the left parotid gland, which proved to be a benign lymphoepithelial lesion (BLEL) associated with Sjogren syndrome preceding the appearance of the MALT lymphoma [1]. Differential diagnosis: Tumor-like benign lymphoepithelial lesion (BLEL), also known as lymphoepithelial sialadenitis associated with Sjogren syndrome; HIV-related benign lymphoepithelial lesion [1].
THERAPY PLANNING:
Differentiation between BLEL and MALT lymphoma is often difficult, but nowadays can be made using Dynamic Contrast-Enhanced MRI (for BLEL, time to peak is more than twice compared with MALT lymphoma) and Diffusion-weighted MRI (ADC values are lower in case of BLEL, compared to MALT lymphoma). Differentiation from HIV-related benign lymphoepithelial lesions is based on the fact that solid masses of the parotid gland are always associated with cystic masses, tonsillar hyperplasia, and cervical reactive adenopathy [2,3].
OUTCOME:
Lymphomas associated with Sjogren Syndrome often present as an indolent lymphoma with rare involvement of the bone marrow and absence of B symptoms. Only in 10% of the cases, low-grade lymphomas can transform into high-grade lymphoma, most frequently Diffuse Large B-Cell Lymphoma [4].
PROGNOSIS:
MALT lymphoma associated with Sjogren Syndrome tends to have a good prognosis and can be cured with surgical excision. When surgical treatment is not an option, chemotherapy and radiotherapy can be considered [5].
5Teaching Points
1. Patients with Sjogren syndrome present an increased risk of developing lymphoma (approximately 7-19 times higher compared with the generally healthy population), especially MALT subtype.
2. Ultrasound is the imaging modality of choice in the follow-up of patients with Sjogren Syndrome, but in case of suspicion of lymphoma should be completed with MRI.
3. Ultrasound and DCE-MRI can help in the differentiation between BLEL and MALT lymphoma in patients with Sjogren Syndrome.
2. Ultrasound is the imaging modality of choice in the follow-up of patients with Sjogren Syndrome, but in case of suspicion of lymphoma should be completed with MRI.
3. Ultrasound and DCE-MRI can help in the differentiation between BLEL and MALT lymphoma in patients with Sjogren Syndrome.
6References
1. Saha D, Tapadia R, Lobo FD, Dhavalpure N, Swamy M, Murali N. Lymphoepithelial sialadenitis involving HIV-Infected and Sjogren syndrome patients: A cytologic study. Indian J Otolaryngol Head Neck Surg. 2019 ;71(2):176-181.
2. Zhu L, Zhang C, Hua Y, et al. Diffusion weighted-MRI in distinguishing between tumor-like benign lympho-epithelial lesion and mucosa-associated lymphoid tissue lymphoma of parotid gland. Int J Clin Exp Med 2016;9(3):6295-6301.
3. Zhu L, Zhang C, Hua Y, et al. Dynamic contrast-enhanced MR in the diagnosis of lympho-associated benign and malignant lesions in the parotid gland. Dentomaxillofac Radiol. 2016;45(4):20150343.
4. Nocturne G, Mariette X. Sjögren Syndrome-associated lymphomas: an update on pathogenesis and management. Br J Haematol. 2015;168(3):317-27.
5. Baik J, Baek HJ, Ryu KH, An HJ, Song S, Lee HJ, Kang Y. MALT lymphoma of the tongue in a patient with Sjögren's syndrome: A case report and literature review. Diagnostics (Basel). 2021;11(9):1715.
2. Zhu L, Zhang C, Hua Y, et al. Diffusion weighted-MRI in distinguishing between tumor-like benign lympho-epithelial lesion and mucosa-associated lymphoid tissue lymphoma of parotid gland. Int J Clin Exp Med 2016;9(3):6295-6301.
3. Zhu L, Zhang C, Hua Y, et al. Dynamic contrast-enhanced MR in the diagnosis of lympho-associated benign and malignant lesions in the parotid gland. Dentomaxillofac Radiol. 2016;45(4):20150343.
4. Nocturne G, Mariette X. Sjögren Syndrome-associated lymphomas: an update on pathogenesis and management. Br J Haematol. 2015;168(3):317-27.
5. Baik J, Baek HJ, Ryu KH, An HJ, Song S, Lee HJ, Kang Y. MALT lymphoma of the tongue in a patient with Sjögren's syndrome: A case report and literature review. Diagnostics (Basel). 2021;11(9):1715.